BASH Guideline
Section 1: The Clinical Approach
Section 2: Primary Headaches

2.1. Migraine
2.1.3 Management
A) General principles
Validate the impact the condition has on the individual and family.
Manage expectations: Patients may have low or unrealistic expectations of what is achievable. Explain that migraine cannot be cured but can be effectively managed in most cases (Gaul et al., 2016; Rothrock et al., 2006).
Reassurance: Patients often worry about an underlying serious disorder. Explaining the nature of the condition to the patient can be of therapeutic value (Laughey et al., 1999).
Empower the patient to help promote self-management (Kindelan-Calvo et al., 2014). Signpost to patient resources such as the Migraine Trust.
Prescribing decisions should be made with reference to the patient’s current clinical situation and their future plans (e.g. pregnancy or contraception). Potential issues of medication overuse, both with respect to the impact on headache and side effects should be discussed.
B) Acute Treatment
Acute treatment choice should be based on the severity of the attack. Factors to consider include how quickly the symptoms develop, presence of significant nausea or vomiting, drug side effects and contraindications and if the patient is pregnant.
Acute treatments can be associated with the development of medication overuse headache (Limmroth et al., 2002). Acute treatments should be restricted to 8-10 days per month to prevent the development of Medication Overuse Headache. Opioids are not recommended for the treatment of acute headaches because of the significant risk of medication overuse and the most protracted withdrawal (Limmroth et al., 2002).
Adding an anti-emetic to an acute treatment improves efficacy unrelated to nausea and/or vomiting (Ross-Lee et al., 1983) and can improve gastric motility and hence drug absorption (MacGregor et al., 1993; Tokola, 1988; Tokola et al., 1983).
The end point of an effective treatment is a significant response at two hours, because the natural history for most attacks is to spontaneously improve in 4 hours (Edmeads, 2005).
If a treatment is not effective at 2 hours, then it is unlikely to work in that attack at that dose and considering an alternative acute treatment or combination treatment would be reasonable (Dahlöf, 2006).
For mild to moderate attacks consider Aspirin 900 mg or ibuprofen 400-600 mg. If NSAID are contra-indicated or not tolerated consider paracetamol 1000mg.
For moderate to severe attacks consider a triptan if there is no contraindication. If a patient can’t take a triptan due to contraindications, side effects or if simple analgesia does not work, rimegepant can be considered.
Triptans are most effective when taken early in the headache phase of the attack (Derry et al., 2014).
Triptans are less likely to be effective at treating the headache if taken during the preceding aura (Bates et al., 1994; Derry et al., 2012; Olesen et al., 2004).
After 2 treatment failures with a particular triptan, a trial with an alternative triptan is recommended. This rationale is based on the finding that in patients who experienced treatment failure in two attacks, 70% failed to respond in the third attack. Around 30% patients do not respond to any triptan (Dahlöf, 2006). Lack of response to one triptan does not predict response to other triptans (Dahlöf, 2006).
SIGN Guideline 155 highlights that the risk of serotonin syndrome combining a triptan with a SSRI/SNRI antidepressant is very rare and in clinical practice they can be used together.
For patients attending the emergency department parenteral NSAIDs or subcutaneous sumatriptan should be considered, and evidence also supports the use of antiemetics (Bigal et al., 2002). Opioids have not been shown to be significantly effective and should not be used (Marmura et al., 2015).
C) Choosing a triptan
Sumatriptan 6 mg subcutaneous remains the most rapid and effective treatment but has a higher risk of adverse events than other formulations. It is a good option in patient who vomit early in their migraine attack.
Nasal Zolmitriptan has moderate nasal absorption and can be beneficial if significant nausea. Nasal Sumatriptan has minimal nasal absorption and has the same NNT as sumatriptan 100mg. All melt in the mouth triptans are gastrically absorbed and have no significant buccal absorption.
Combination of a triptan and an NSAID with a long half-life, such as naproxen, is better than monotherapy (Law et al., 2016).
In comparison to sumatriptan 100 mg (Cortelli et al., 2011; Evers et al., 2015; Ferrari et al., 2001; Poolsup et al., 2005):
- Lower adverse events: naratriptan 2.5mg, almotriptan 12.5mg and frovatriptan 2.5mg
- Better 2-hour pain response: eletriptan 80mg and rizatriptan 10mg, almotriptan 12.5mg
- Lower recurrence rate: frovatriptan 2.5mg, and eletriptan 40mg
Contraindications to triptans include ischaemic heart disease, cerebrovascular disease, previous myocardial infarction, and uncontrolled or severe hypertension. The cardiovascular risk of triptans is very low in the absence of these contra-indications (Dodick et al., 2004).
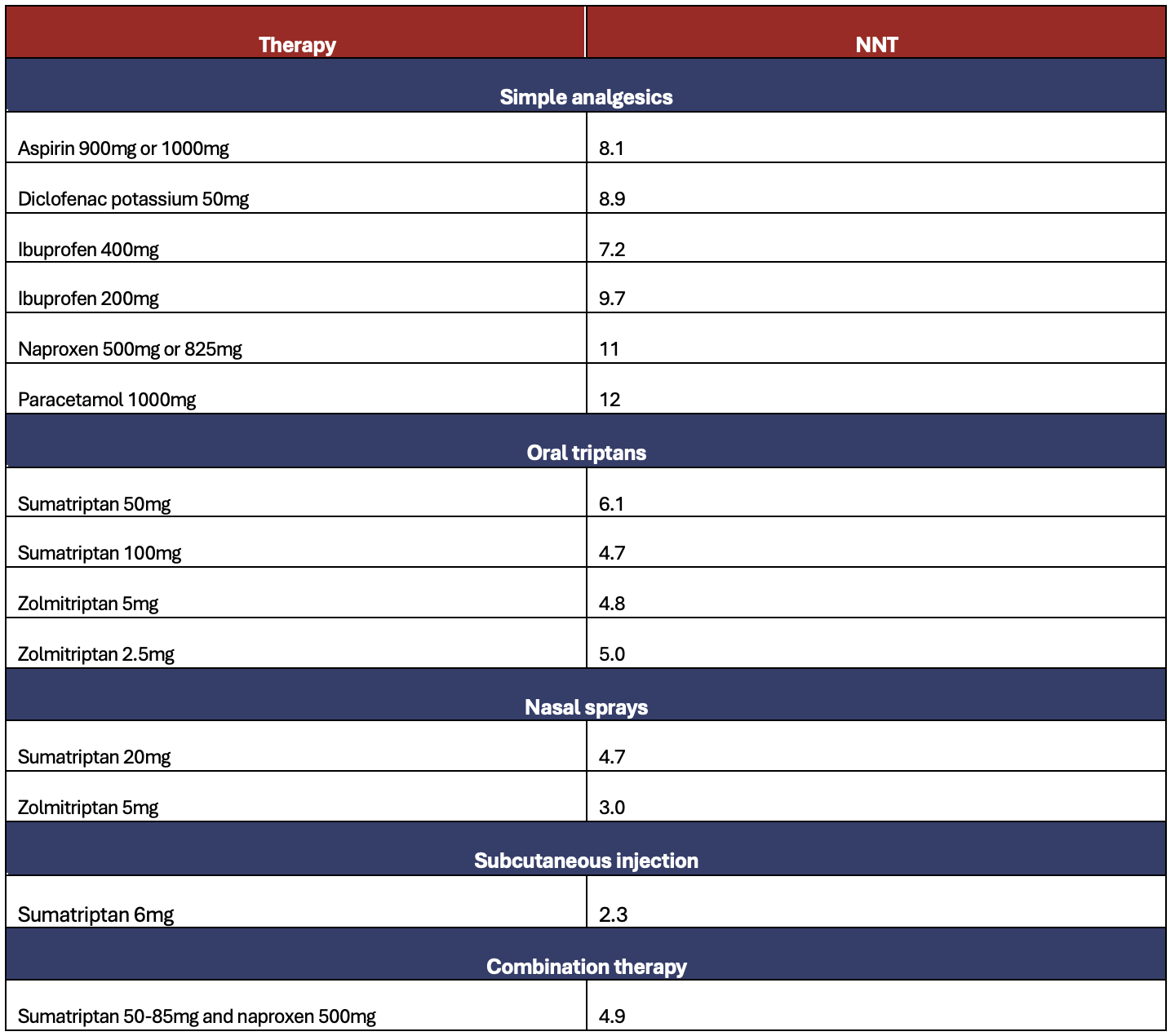
The NNTs for therapies to achieve the outcome of pain freedom at two hours from a baseline of moderate to severe pain has been documented by the Scottish Intercollegiate Guidelines Network (SIGN 155 February 2018), with information collated from the Cochrane reviews (Table 2).
Table 2: Calculated numbers needed to treat for acute migraine therapies for an outcome of pain free at two hours in patients with moderate to severe pain, compared to placebo.
Adapted from the Scottish Intercollegiate Guidelines Network, Pharmacological management of migraine guidelines (SIGN 155 February 2018) https://migrainetrust.org/
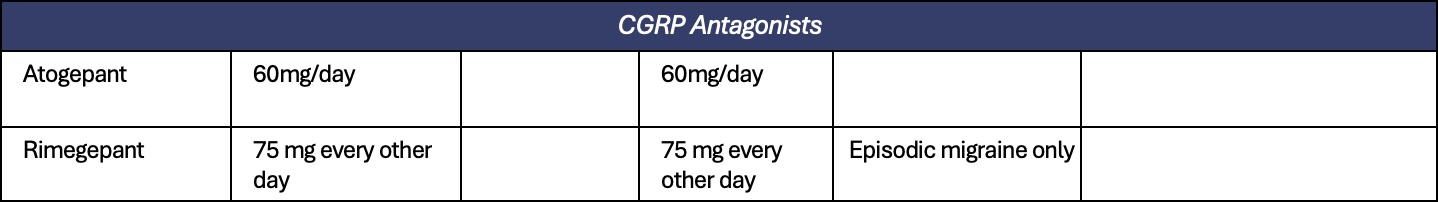
D) Gepants (CGRP antagonists)
Gepants (Rimegepant) is recommended as an option for the acute treatment of migraine with or without aura in adults who have tried at least 2 triptans and they did not work well enough or there is contraindication or intolerance to triptan where nonsteroidal anti-inflammatory drugs (NSAIDs) and paracetamol were tried and did not work well (Croop et al., 2019; Lipton et al., 2019). Contraindications to triptan include ischaemic heart disease, cerebrovascular or peripheral vascular disease and uncontrolled hypertension.
Recommended acute treatments are included in tables 3, 4 and 5.
Table 3. Recommended acute treatments – simple analgesics and antiemetics
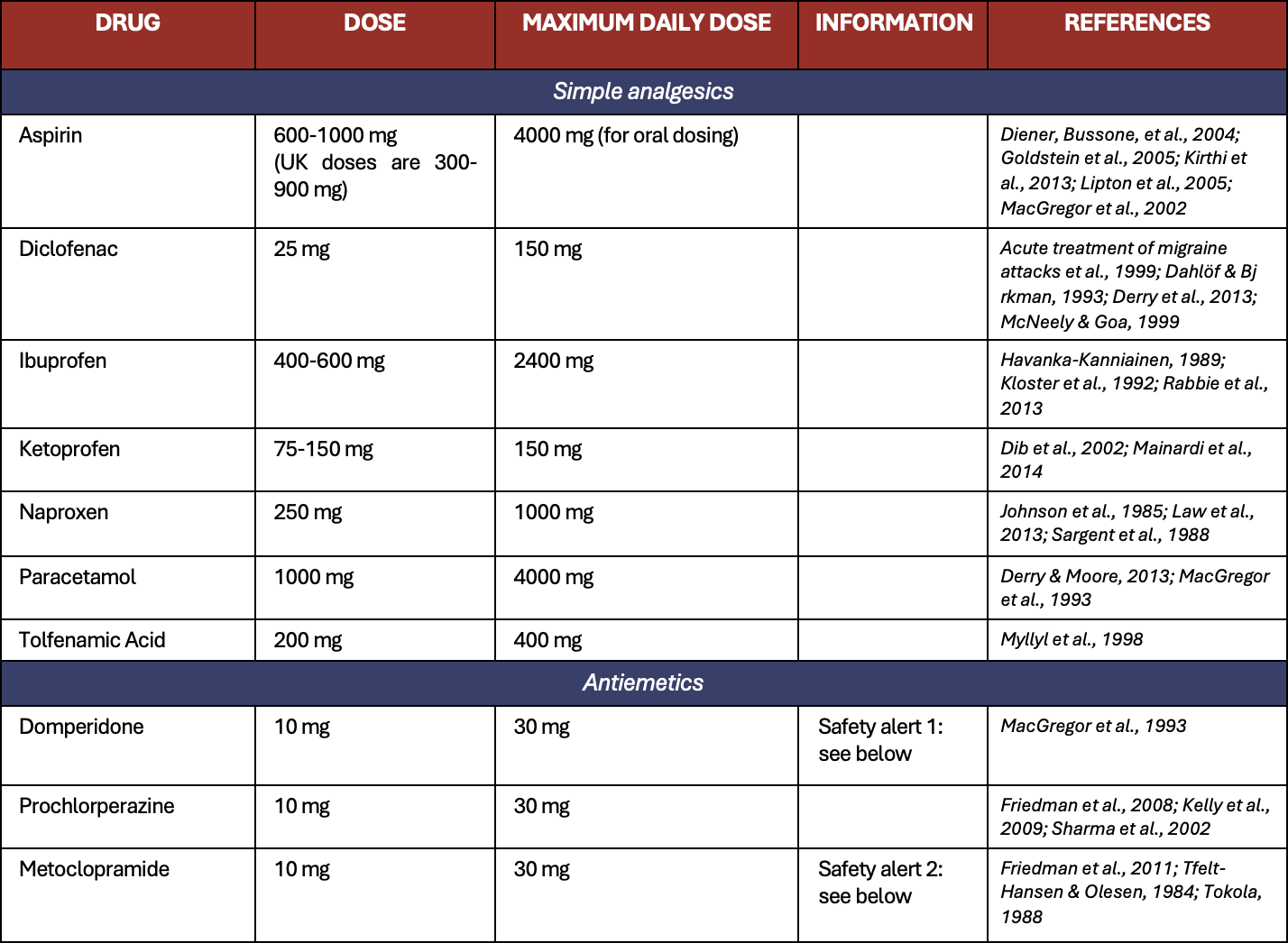
Table 4. Recommended acute treatments – Triptans
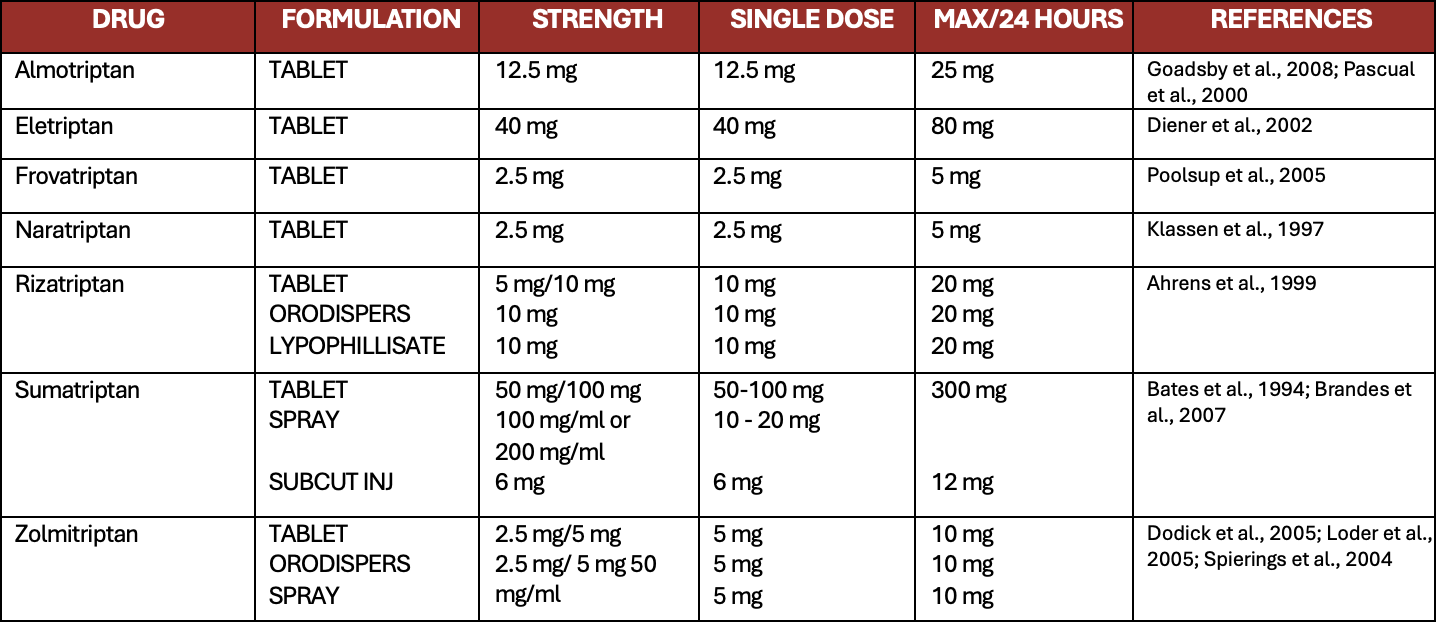
Table 5. Recommended acute treatments - Gepants

E) Preventative treatment
General principles
The aim of preventive treatment is to reduce the frequency, severity and duration of migraine attacks and to reduce the risk of medication-overuse headache.
The decision to start preventive treatment should be a joint decision with the patient and made with reference to the patient’s current clinical situation and their future plans (e.g. pregnancy or contraception).
NICE recommends preventive treatment should be considered if
- Migraine attacks have a significant impact on quality of life and daily function; for example, they occur frequently (more than once a week on average) or are prolonged and severe despite optimal acute treatment.
- Acute treatments are either contraindicated or ineffective.
- The person is at risk of medication overuse headache (MOH) due to frequent use of acute drugs.
Such an approach will also mitigate the risk of escalation of acute treatment and consequent development of medication overuse headache.
Acute treatment on more than 2 days per week is associated with medication overuse, which renders preventive treatment less effective (Zeeberg et al., 2006a).
As there are relatively few head to head comparative studies, the choice of preventive depends primarily upon the side-effect profile of the drug and co-existing morbidities.
Preventive medications, without a fixed dose, should be started at low dose and be titrated every 1-2 weeks as tolerated to an effective or maximum tolerable dose and continued for at least 6-8 weeks to adequately assess effect (Albsoul-Younes et al., 2004; Diener, Agosti, et al., 2007). It is important that patients are reviewed regularly in order to stop patients being left in limbo on small ineffective doses.
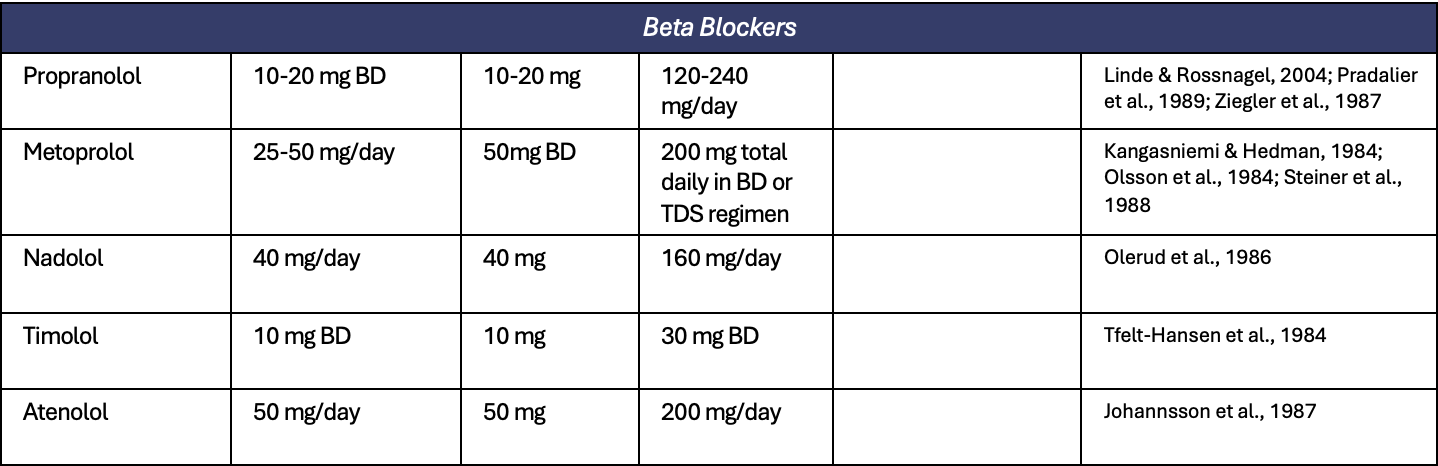
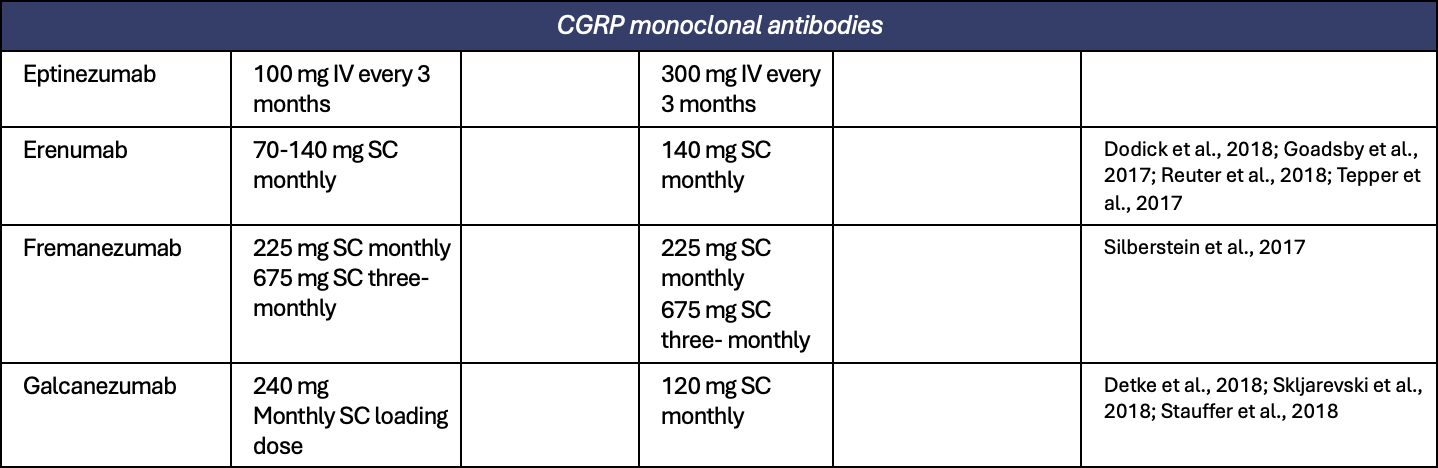
Targeted treatments against CGRP can be used if three standard preventatives, namely amitriptyline/nortriptyline, candesartan, propranolol or topiramate have been considered. Note a patient does not have to have been prescribed 3 preventers if there are contra-indications to a particular medicine e.g. betablocker and asthma, topiramate and a female of childbearing age.
The aim of preventer treatment is to reduce migraine days to 4 or less a month.
For some patients, improvement in quality of life may be a better marker of treatment success than an absolute measure of number of days.
A headache diary may help evaluate response to treatment.
In patients showing a response to treatment, continuation of preventative therapy is recommended for at least 12 months at which point a review is suggested.
Treatment options
In selecting a preventative treatment, a reasonable strategy would be to consider which options might be most suitable for the individual patient, given their previous treatment, medical and other co-morbidities, personal preferences, and side effect profiles of the various treatments.
Table 6 shows the dose and titration regimen for recommended preventive treatments in both episodic and chronic migraine.
Table 7 shows the dose and titration regimen for recommended preventive treatments in episodic migraine only.
Table 8 shows dose and treatment regimen for recommended preventive treatments in chronic migraine only.
All preventive treatments with randomised placebo-controlled trial data are listed as an appendix.
Table 6. Recommended preventive treatments in episodic and chronic migraine.
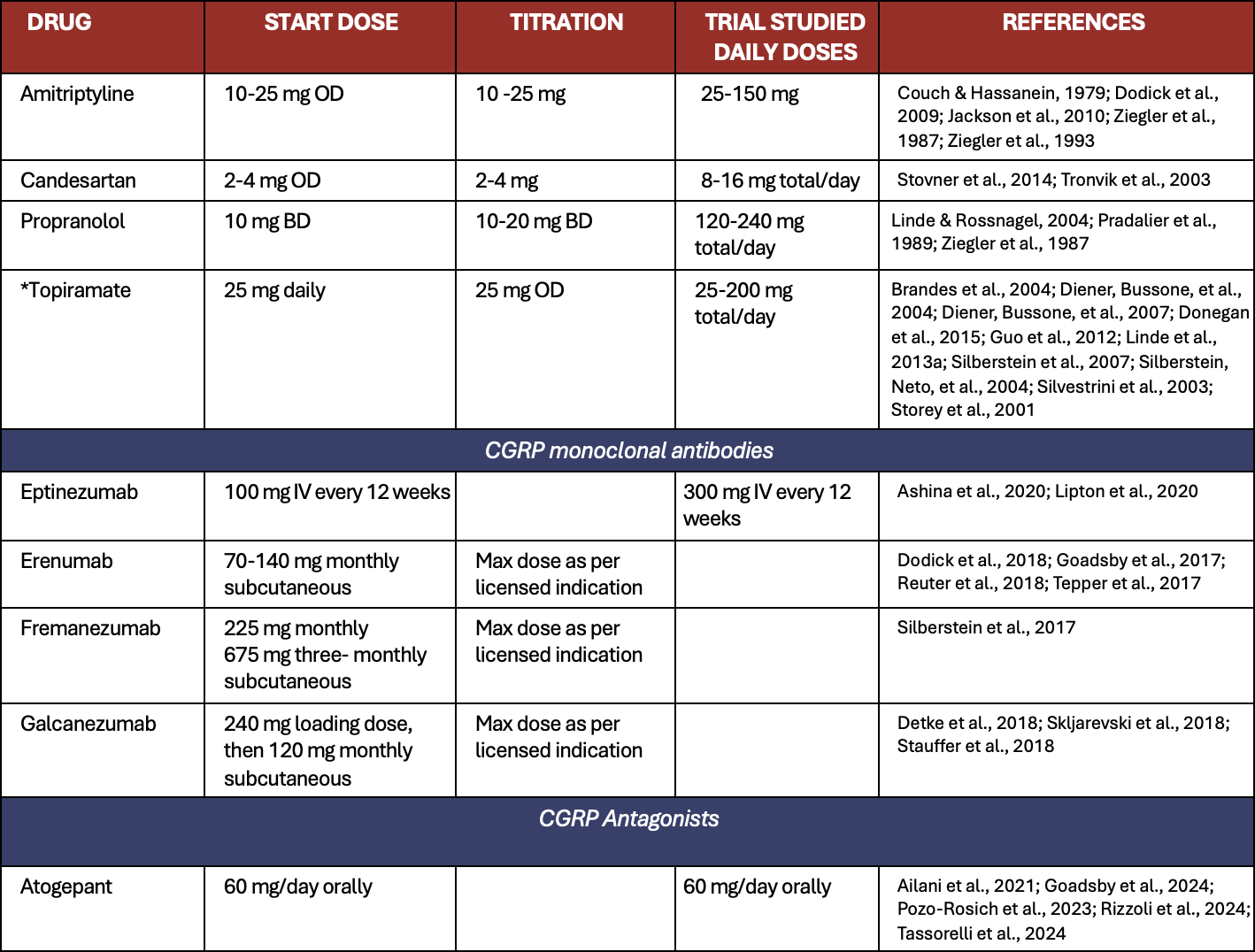
*Topiramate Patient Safety Alert
Use of topiramate during pregnancy is associated with significant harm to the unborn child. Harms included a higher risk of congenital malformation, low birth weight and a potential increased risk of intellectual disability, autistic spectrum disorder and attention deficit hyperactivity disorder in children of mothers taking topiramate during pregnancy. Topiramate is now contraindicated in pregnancy and in women of childbearing potential unless the conditions of a pregnancy prevention program are fulfilled.
Accessed 28 July 2025
Table 7. Recommended Preventive Treatments for Episodic Migraine Only
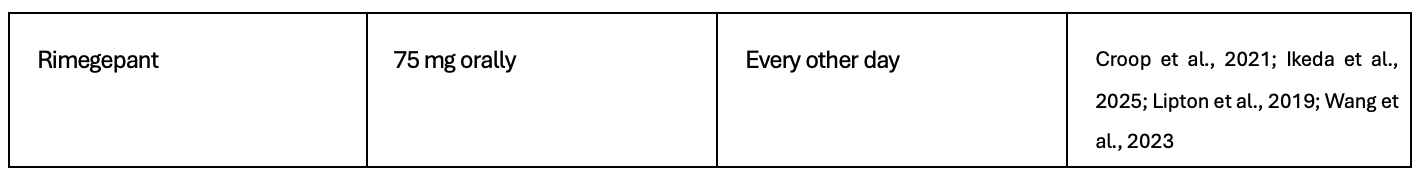
Table 8. Recommended preventive treatments for chronic migraine only

*Valproate Patient Safety Alert
In girls and women of childbearing potential, valproate should be initiated and supervised by a specialist and only prescribed when other medications have not been tolerated or have found to be ineffective. This is because of 30-40% risk of neurodevelopmental disability in unborn babies exposed to valproate (MHRA April 2017). Valproate should only be prescribed by following the MHRA guidance, including a signed contraceptive plan and signed consent form documenting discussion of the risks (see MHRA website)
https://www.gov.uk/drug-safety-update/valproate-and-developmental-disorders-new- alert-asking-for-patient-review-and-further-consideration-of-risk-minimisation-measures
F) Menstrual Migraine
A proportion of women suffer from migraine attacks in association with the menstrual cycle, termed menstrual related migraine (MRM). MRM occurs between days -2 and +3 of the first day of menstruation (which is +1) in at least 2 out of 3 menstrual cycles.
Women with MRM will also have attacks at other times.
Less than 10% of women report migraine exclusively with menstruation and at no other time (‘pure’ menstrual migraine) (Couturier et al., 2003; Granella et al., 1993; MacGregor et al., 1990; Stewart et al., 2000; Vetvik et al., 2010; Wöber et al., 2007).
Acute Treatment
The acute treatment of menstrual related attacks is no different to non-menstrual attacks.
Head-to-head studies do not show clear superiority of one triptan over any other (Mett & Tfelt-Hansen, 2008).
Recommended short term preventive treatments for menstrual related migraine, or pure menstrual migraine.
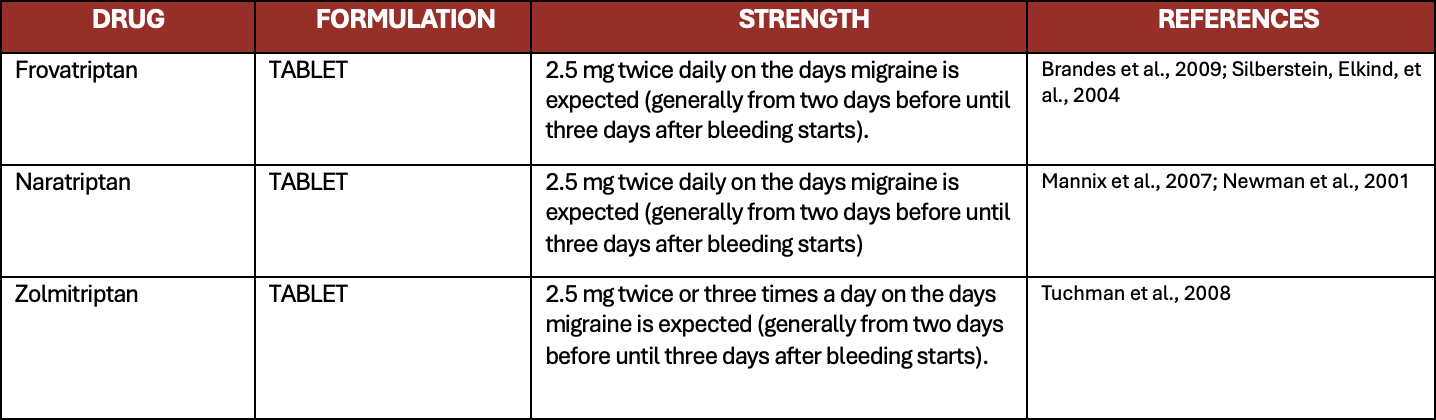
Table 9. Recommended triptans for short term prevention of menstrual related migraine or pure menstrual migraine

Targeted oestrogen supplementation
Menstrually targeted oestrogen supplementation (assuming no contraindications) has been found in some studies to offer benefit in menstrual related migraine (de Lignières et al., 1986; Dennerstein et al., 1988; Pradalier et al., 1994).
However, a rebound increase in migraine attack frequency has been found when the effect of this strategy has been considered over the whole menstrual cycle (MacGregor et al., 2006).
The risk of stroke in migraine with aura, when taking oestrogen-containing contraceptives
Females suffering migraine with aura have an inherent increased risk of stroke (Kurth et al., 2005).
Use of the oestrogen contraceptive pill is also associated with increased risk of stroke in all individuals. The incidence of stroke in females with migraine with aura, who are also taking the oestrogen-containing contraceptive pill is additionally increased.
Consequently, contraceptive methods other than oestrogen containing contraception are advised for women with migraine with aura. There is no established additional risk in migraine without aura.
G) Treatment in pregnancy & breast feeding
In the majority of women, migraines improve during pregnancy (Granella et al., 2000; Sances et al., 2003)
Caution is advised and checking with British National formulary data and pregnancy register is recommended especially when prescribing in pregnancy, breast feeding, and considering contraception. The resource Best Use of Medicine in Pregnancy (BUMPS) may also be of help to patients (http://www.medicinesinpregnancy.org/).
Paracetamol is not generally considered to be associated with a significantly elevated risk throughout pregnancy and lactation (MacGregor, 2014)
The Sumatriptan & Naratriptan Pregnancy Registry found no evidence of teratogenicity associated with major birth defects for sumatriptan (Ephross & Sinclair, 2014; Marchenko et al., 2015; Nezvalov -Henriksen et al., 2013; Spielmann et al., 2018)
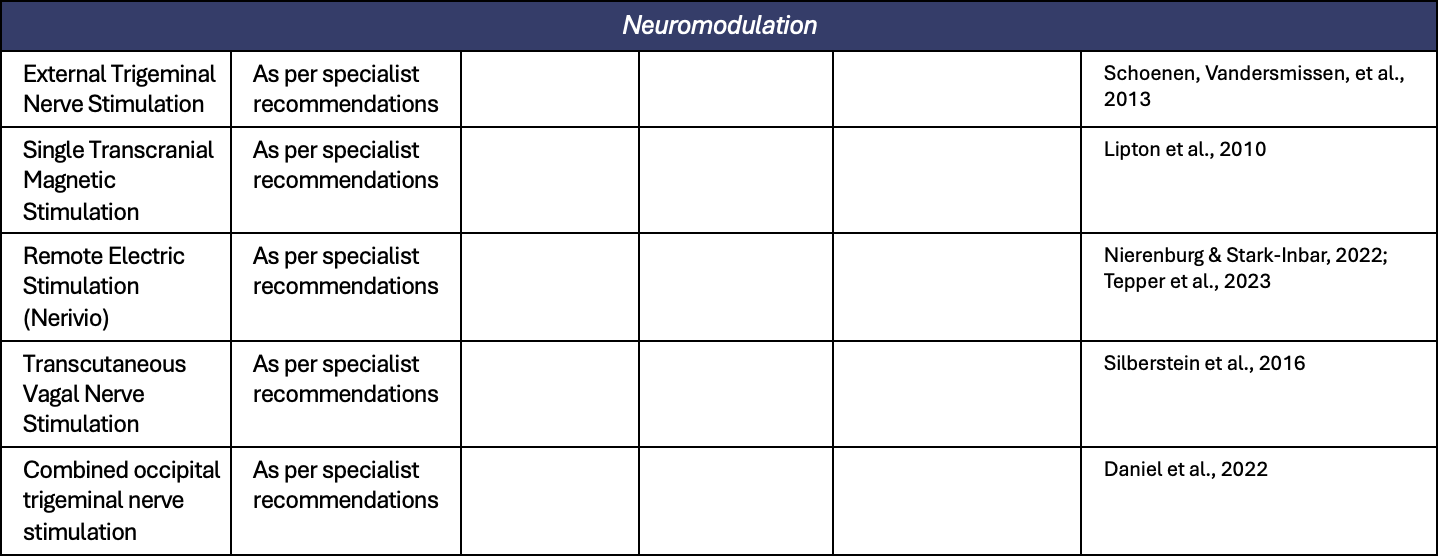
Appendix 1. All preventive treatments for migraine
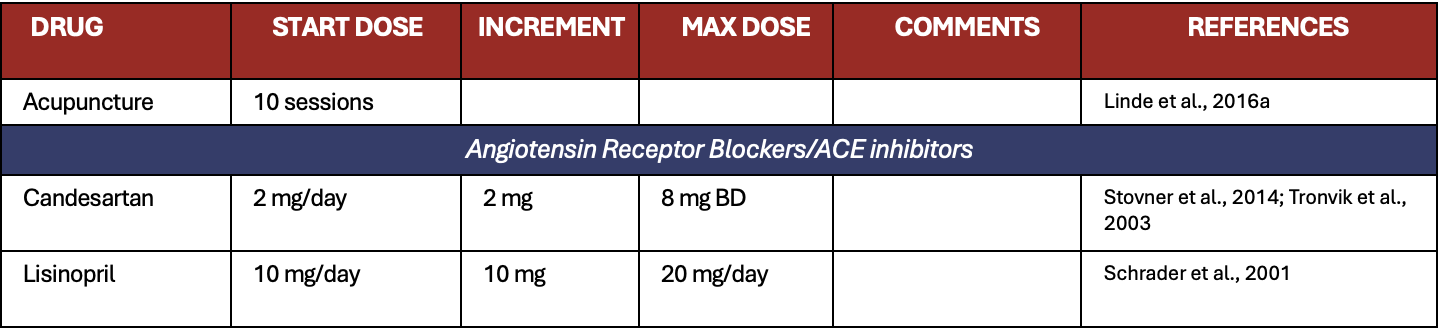
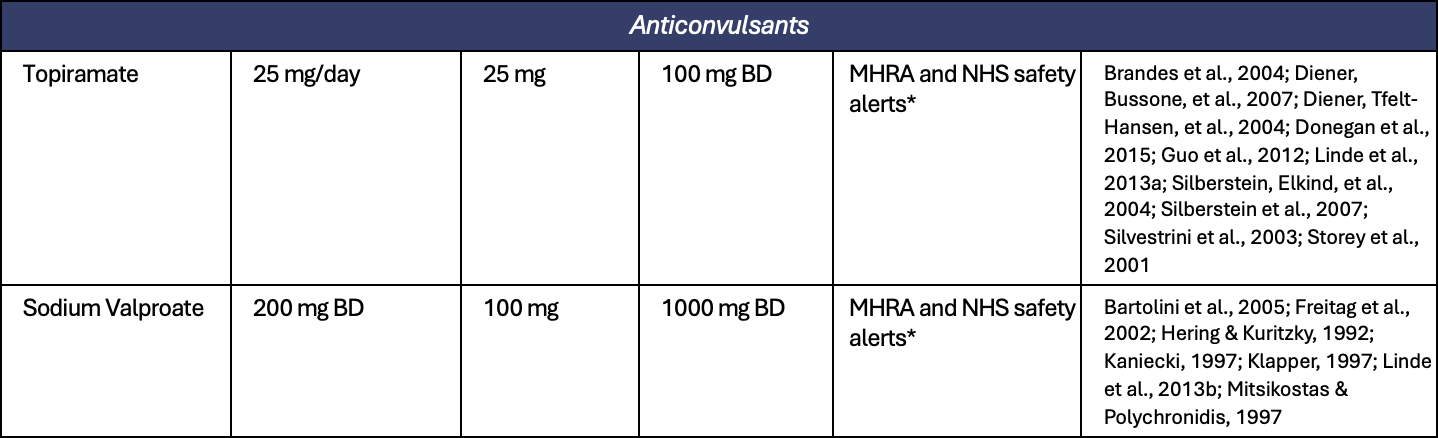


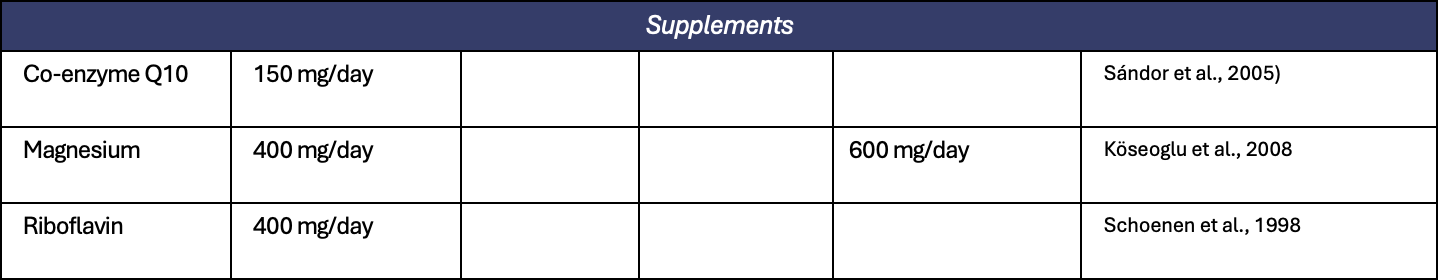

Appendix 2. All treatments for short term prevention of menstrual related migraine or pure menstrual migraine